The race to bring down coronavirus began almost as soon as it escaped Wuhan. Nine months on, research to develop a vaccination for COVID-19 has gotten a lot closer.
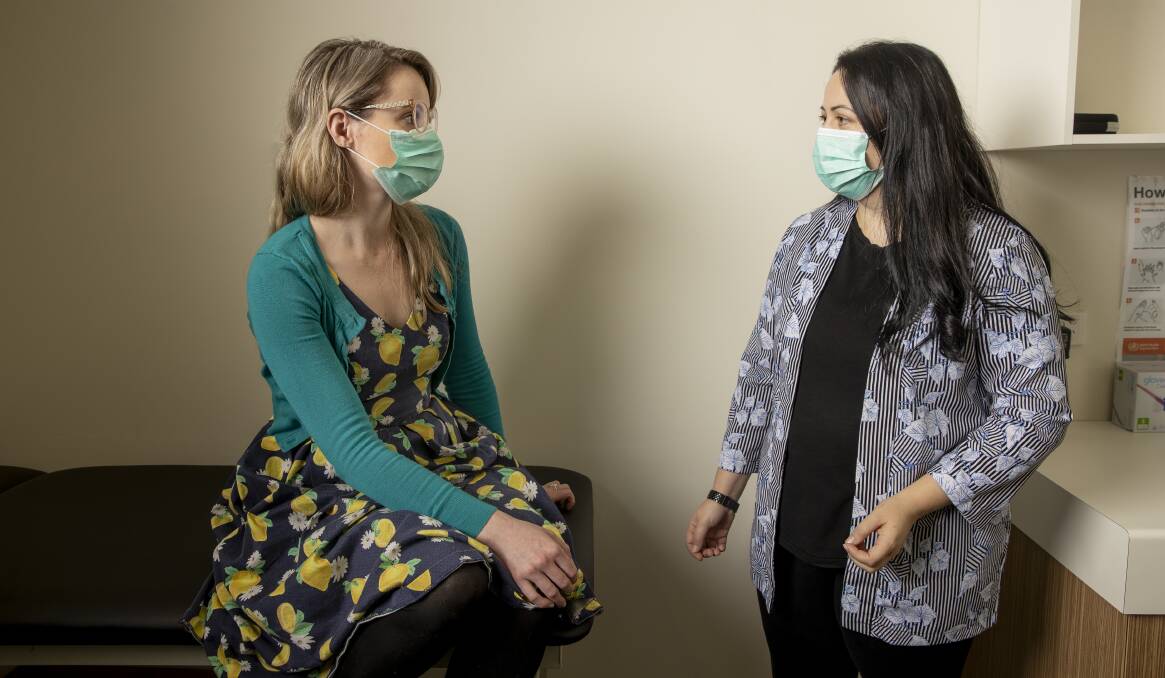
Inside the Ochre Health Hub at the University of Canberra this week, Tamara Hughes was due to receive her second dose of a potential vaccine developed by United States company Novavax, one of several trials showing promising enough results to proceed past initial testing.
The biotechnology company recruited 1500 people from 40 locations across the United States and Australia after its early trial found high levels of neutralising antibodies in phase one volunteers.
Around 80 Canberrans received their second shot at the Bruce campus research facility this week - all going according to plan - the final jab will be administered in five months time.
One of the Paratus Clinical researchers overseeing the study, Matt Clacy, is part of a team which will meet with Canberra participant 10 times over the next year.
Mr Clacy said exposure to the vaccine - a small component of the virus - trains the body's immune system to recognise COVID-19 and respond accordingly.
He said participants weren't at risk of being infected with COVID-19 from the trial vaccine and should not feel any coronavirus symptoms.
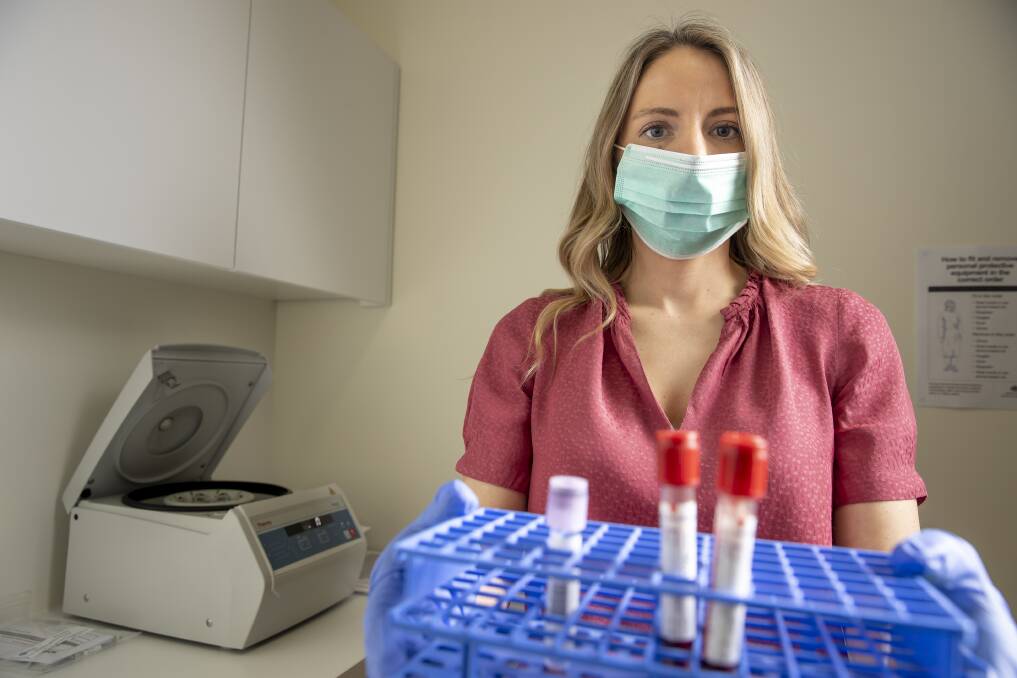
The volunteers will have blood taken each time they attend the facility, which is then tested for detection of antibodies to COVID-19.
Last month, Novavax flagged its intention to start the next large-scale trials in September, with the hope of manufacturing up to two billion doses of the vaccine in 2021.
Mr Clacy predicts there will be a vaccine ready for distribution in the first half of next year.
"But that's just my own personal speculation," Mr Clacy said.
As was the case recently with the Oxford University trial, Mr Clacy said it wasn't uncommon for trials to be interrupted if participants felt unwell for any reason.
Since resumed, the Oxford trial has been heralded as one of the most promising among those currently underway.
There are currently 182 COVID-19 vaccine candidates in development including 36 in human clinical trials.
A spokesperson for the Department of Health said the Australian government was monitoring the progress of each one, including the Novavax vaccine, one of four human clinical trials under way in Australia.
To date, the government has committed $362 million to vaccine development, including $25 million to support trials in Australia.
The Department of Health spokesperson said negotiations were ongoing and were commercial in confidence.
Mr Clacy said while there was no doubt competition from companies to get their product to market first, it was by no means an international scrap.
"There's a great degree of transparency between bodies, be they commercial, governmental or not-for-profit institutes," he said.
"The collaboration and willingness to share information has been greater than I have ever seen in my time within clinical research."
Mr Clacy said it would be remiss of him to speculate on which would likely be the most successful vaccine.
"What I will say is there's a number of different vaccines being tested and they're working off different modes of action," Mr Clacy said.
"So the positive thing is we're not all just going down one pathway, there's a number of pathways being explored."
Ms Hughes, 30, said whether or not Novavax won the vaccine race was irrelevant, the more healthy people able to contribute to the research, the quicker the return to life pre-coronavirus.
"My mum has lupus and has significantly compromised lung function," Ms Hughes said.
"She's an older lady who already has trouble socialising because she gets quite tired easily.
"Coronavirus has meant that she has to think twice about going out to see people, you can't go in for the big hug and she's reluctant to go anywhere there might be crowds."
READ ALSO:
Ms Hughes said her only concern in volunteering to take part was the idea that if a different trial proved successful first she would be prohibited from receiving it.
"I was assured that would not be the case," she said.
"When a vaccine is finally available there is going to be unprecedented demand.
"Perhaps this vaccine won't be the first one available but it means we've got choice, it means this vaccine might work well for certain populations better than others so any research in this space is always going to be really valuable."


